More than a year since the Coronavirus completely changed our lives, we are currently in the phase of gradually moving toward immunity. In coordination with the World Health Organization (WHO), different groups have been working toward the development of COVID-19 vaccines to combat the pandemic. While it took some time to develop, its production has ultimately helped protect the people and control the spread of the virus.
Understanding COVID-19 Vaccines
How do vaccines work?
Vaccines work by preparing the body’s immune system to recognize and fight off the viruses present. Vaccines contain material from a specific virus—mimicking the virus that causes the disease. This will trigger the body’s creation of antibodies. After vaccination, these antibodies will provide protection once a person is infected with the actual virus. It usually takes two weeks after vaccination for the body to build protection against the virus.
Who should get vaccinated?
Different COVID-19 vaccines are generally safe for ages 18 years and older. Even those who have pre-existing conditions can receive the vaccine. These conditions include hypertension, diabetes, asthma, pulmonary, liver and kidney disease. Nevertheless, it is still important to discuss your health conditions with a health professional and vaccine provider.
Who should not get vaccinated?
People with severe allergic reaction (anaphylaxis) or immediate allergic reaction to any component of the vaccine should not take the vaccine. In any instance that you had an allergic reaction after getting the first dose, you should not get the second dose.
A severe allergic reaction needs to be treated with an epinephrine or medical assistance. On the other hand, immediate allergic reaction includes hives, swelling, or wheezing. These symptoms manifest within 4 hours after getting the shot. Seek medical assistance if you experience allergic reactions.
Additionally, there is an age group covered by Food and Drug Administration (FDA) approval for each vaccine. It will be helpful to read and follow guidelines before receiving any authorized COVID-19 vaccine.

Different COVID-19 Vaccines
Currently, there are different vaccines available, all of which being guaranteed safe and effective. These vaccines differ in their composition and how they trigger the creation of antibodies to protect the body from the virus. Below are some of the vaccines that has been evaluated and authorized by the World Health Organization (WHO).
1. Oxford-AstraZeneca
Manufacturer: AstraZeneca
Type of Vaccine: Viral vector
Recommended dosage:
The AstraZeneca vaccine is given in two (2) shots, 0.5 ml each. The dosage is administered intramuscularly with an interval of 8 to 12 weeks.
Who are eligible to get the vaccine?
The AstraZeneca vaccine is recommended for people aged 18 and older. Medical experts continue clinical trials to determine its safety and protection for persons aged more than 56 years old.
Effectiveness:
Based on clinical trials, the efficacy of the AstraZeneca vaccine is up to 70.4% against symptomatic SARS-COV-2 infection. It is also 100% effective against severe COVID-19. Nonetheless, longer dose intervals within 8 to 12 weeks have been associated with greater efficacy.
2. Johnson and Johnson-Janssen
Manufacturer: Janssen Pharmaceuticals Companies of Johnson & Johnson
Type of Vaccine: Viral vector
Recommended dosage:
The J&J vaccine is given as one dose, 0.5 ml. The dosage is administered in the muscle of the upper arm. You are considered fully vaccinated 2 weeks after the single dose of J&J vaccine.
Who are eligible to get the vaccine?
The J&J Janssen vaccine is recommended for people aged 18 and older.
Effectiveness
Based on clinical trials, a single dose of J&J Janssen Vaccine was 66.9% effective at preventing COVID-19 infection. Also, it offers ~77%-85% efficacy against severe COVID-19. People achieved most protection 2 weeks after vaccination.
3. Moderna
Manufacturer: ModernaTX, Inc.
Type of Vaccine: mRNA
Recommended dosage:
It is recommended to receive the Moderna vaccine in two doses, 0.5 ml each, at an interval of 28 days apart. Likewise, the vaccine is administered in the muscle of the upper arm. The interval between the doses may be extended to 42 days. Although, some reports show that with vaccine supply constraints, the second dose can be delayed up to 12 weeks.
Who are eligible to get the vaccine?
The Moderna vaccine is recommended for people aged 18 and older
Effectiveness:
The Moderna vaccine showed an efficacy of 94.1% in clinical trials, starting 14 days after the first dose. It offers 100% efficacy against severe COVID-19.
4. Pfizer-BioNTech
Manufacturer: Pfizer, Inc. and BioNTech
Type of Vaccine: mRNA
Recommended dosage:
The Pfizer-BioNTech COVID-19 vaccine is administered in two (2) shots, 0.5 ml each. The dosage is administered intramuscularly in an interval of 21 days apart.
Who are eligible to receive the vaccine?
The Pfizer-BioNTech vaccine is recommended for people ages 5 years and older.
Effectiveness:
Based from clinical trials, Pfizer-BioNTech vaccine was 95% effective in people ages 16 years and older. While in children ages 5-15 years old, the vaccine is proven to have > 90% efficacy. The immune response of children ages 5-15 years was at least as strong as that of people aged 16-25.
5. Sinovac-CoronaVac
Manufacturer: Sinovac
Type of Vaccine: Inactivated virus
Recommended dosage:
Vaccination with Sinovac-CoronaVac is recommended in two (2) doses, 0.5 ml each, and is to e administered intramuscularly. WHO recommends Sinovac-CoronaVac to be received at an interval of 2-4 weeks apart. If the second dose is administered less than 2 weeks after the first dose, the dose doesn’t need to be repeated. It should be noted that if the second dose is delayed beyond 4 weeks, it should be given at the earliest possible time.
Effectiveness:
In a large phase 3 trial in Brazil, the vaccine had 51% effectiveness when administered at an interval of 14 days. Additionally, it has an efficacy of 100% against severe COVID-19, and 100% against hospitalization, 14 days after receiving the second dose.
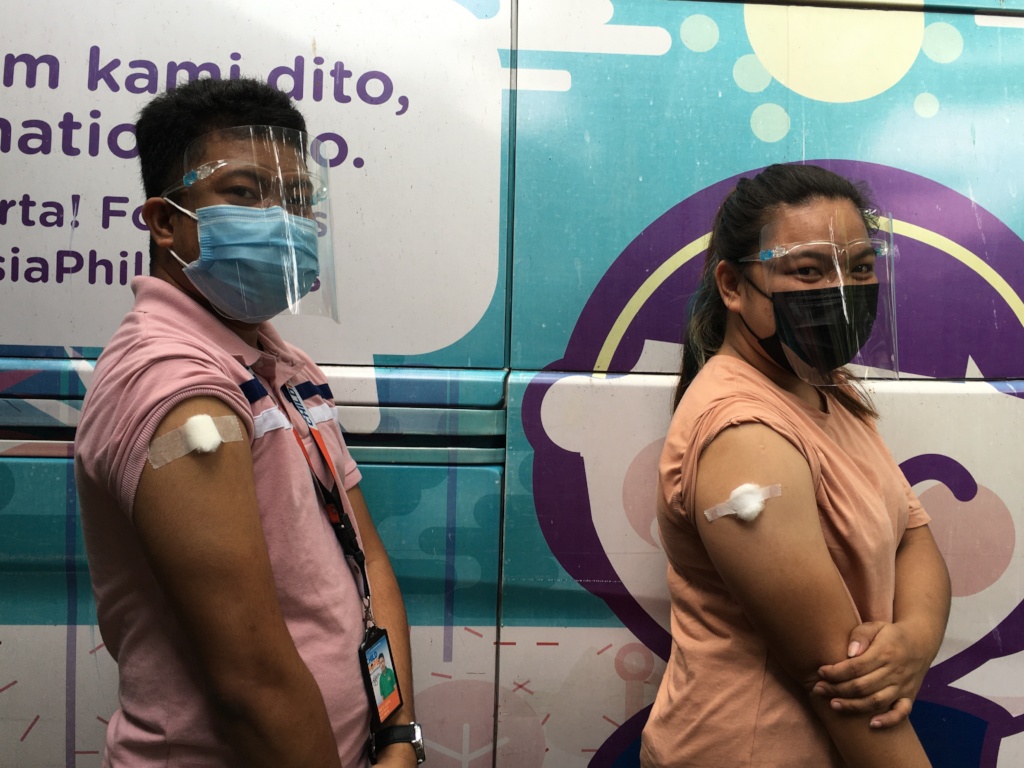
Possible Side Effects of COVID-19 Vaccines Post-Vaccination
Side effects are normal signs that your body is responding to the vaccine and is building protection against the virus. Whatever vaccine you are getting, you can expect one or several of these side effects:
- Tenderness and/or pain at the injection site
- Headache
- General discomfort or malaise
- Muscle pain
- Chills or increased body temperature
- Fever
- Nausea
- Allergic reactions
If side effects or unusual symptoms persist 24 hours after receiving the vaccine, seek medical help or call your provider about taking medication such as paracetamol or antihistamines.
Booster Shot
A COVID-19 booster dose helps improve immunity additional to the first 2 primary doses. It gives you longer-term protection against severe effects of the COVID-19. If you are eligible, you’ll be offered a booster dose at least 6 months after you had your second dose. A booster dose can be any of the authorized COVID-19 vaccine.
Manila Vaccine Registration
According to Statista, approximately 9.1 million are fully vaccinated in the National Capital Region (NCR) as November 2021, while there are still 10.1 million people waiting to receive the second dose. Whereas nationwide vaccination against COVID-19 began last March 2021, the countrywide effort aims to reach 58 million by the end of the year.
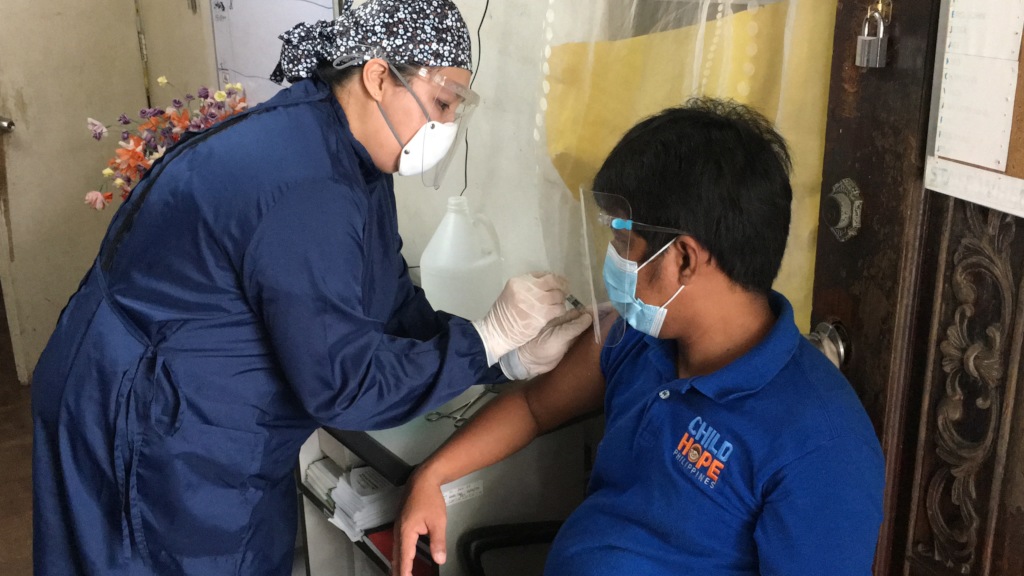
Childhope Street Educators Get Vaccinated Against COVID-19
Reaching out to the street children has been even more challenging during COVID-19. Despite the restrictions, Childhope Philippines still continues its mission of bringing hope to the children and their families through different outreach programs. One way of helping them out is through health education. More than ever, this is the time to raise awareness on the importance of proper healthcare and healthy lifestyle.
Children are among those who are in risk of COVID-19 infection. While they are not eligible for vaccination, the people around them must be prioritized to receive vaccine. These people include teachers and social workers who ensure the well-being of these children and their families. Hence, Childhope staff, street educators, and social workers all got vaccinated to protect, not only themselves, but also the street children.
Childhope Philippines believes that everyone should receive adequate healthcare. This includes equitable and safe access on vaccine. Vaccination is critical to ending the COVID-19 pandemic. With the recent emergence of the Omicron variant, it is very crucial to be vaccinated as soon as the opportunity is available and still observe safety protocols.
